BSA protein functions: why albumin matters in modern biotech
Bovine Serum Albumin (BSA) plays a central role in modern biotechnology, diagnostics, and life sciences. BSA protein functions are widely exploited as stabilizers, blocking agents, and carrier proteins in applications ranging from immunoassays to reagent formulation and protein handling. This versatility has made BSA a long-standing standard across multiple biotech workflows.
However, increasing regulatory pressure, the need for reproducibility, and the shift toward chemically defined systems are challenging the use of animal-derived materials. In this context, recombinant albumin is gaining relevance not merely as an animal-free substitute, but as a solution designed to preserve essential BSA functionality while improving consistency and regulatory alignment across advanced biotech applications.
This shift is accelerating the adoption of recombinant protein platforms designed to ensure functional equivalence, consistency, and regulatory alignment in advanced biotech workflows.
BSA in routine workflows: stabilization, blocking, carrier roles
In routine laboratory and industrial workflows, BSA is used to stabilize enzymes and antibodies, minimize non-specific surface adsorption, transport hydrophobic molecules, and supplement cell culture systems. These BSA protein functions arise from its globular structure, high solubility, and multiple ligand-binding domains.
Traditionally, BSA is purified from bovine plasma through fractionation-based processes. While widely adopted, this approach introduces inherent variability linked to biological raw materials. Differences in animal origin, health status, and processing conditions can affect batch consistency, and even at high purity levels, residual serum proteins or bound compounds may remain.
From a regulatory perspective, animal-derived components require extensive documentation and risk assessment related to biological safety and traceability, including aspects such as transmissible spongiform encephalopathies (e.g., BSE/TSE). As a result, demand for animal-free BSA and robust BSA alternatives has grown, particularly in regulated and high-performance applications.
BSA alternatives: limitations when BSA functions are not fully replicated
Not all alternatives currently available on the market fully replicate native BSA functionality. Some BSA alternatives exhibit altered binding behavior, reduced stabilizing capacity, or lower protection against interfacial stress. These differences can lead to reduced assay robustness, increased signal variability, or the need for protocol re-optimization.
At manufacturing and commercial scale, such limitations complicate validation and increase development costs. In addition, certain alternative products lack a clear regulatory alignment strategy, creating uncertainty during late-stage development or market access. As a result, replacing BSA often remains a compromise rather than a true functional upgrade.
Levprot Bioscience’s rBSA: recombinant albumin engineered to preserve BSA protein functions
Levprot addresses these challenges with an animal-free recombinant albumin engineered to preserve essential BSA protein functions while eliminating risks associated with animal-derived materials. Our solution is developed using precision fermentation proteins, enabling tight molecular control, high reproducibility, and scalability, together with purification processes tailored for regulated biotech applications.
Beyond its animal-free origin, Levprot rBSA has been designed with functional performance as a primary criterion. The protein has been systematically benchmarked against conventional serum-derived BSA under immunoassay conditions, with a specific focus on its behavior as a blocking and stabilizing agent. These evaluations demonstrate that Levprot’s rBSA can achieve equivalent blocking performance while operating at lower working concentrations, maintaining a balanced relationship between specific signal intensity and non-specific background.
This approach allows R&D and diagnostic developers to transition away from animal-derived BSA without compromising assay robustness, while simultaneously improving batch-to-batch consistency and simplifying regulatory alignment. The result is a recombinant albumin solution that fits seamlessly into existing workflows and supports both development and scale-up requirements.
ELISA benchmarking for blocking performance
The functional performance of Levprot’s recombinant BSA has been evaluated using ELISA as a representative and widely accepted model for blocking efficiency and signal control in immunoassays. ELISA provides a sensitive framework to assess surface saturation, non-specific binding, and signal-to-noise balance, all of which are directly linked to core BSA protein functions.
Benchmarking studies show that Levprot rBSA achieves comparable specific signal levels to animal-derived BSA when used at significantly lower concentrations. Equivalent assay performance has been demonstrated at dilution ranges between 1:20 and 1:100, compared to conventional blocking conditions using approximately 1% serum-derived BSA.


Importantly, non-specific background signal remains stable at these reduced concentrations, indicating efficient surface blocking without over-saturation.
Beyond immunoassays, consistent trends reported across multiple external applications indicate that rBSA performance is strongly concentration-dependent in biological systems, particularly in cell culture, stabilization, and cryopreservation workflows. As a highly defined and homogeneous protein, rBSA typically exhibits optimal functional behavior at lower working concentrations than conventional animal-derived BSA, where it acts primarily as a carrier and stabilizing component rather than a source of undefined bioactive factors.
Across different cell-based models, effective performance has been observed at low rBSA concentrations, commonly in the 0.01–0.1% range, supporting cell handling, protection during processing, and post-thaw recovery while reducing the total amount of albumin required. At higher concentrations, the strong surface-coating and binding capacity of rBSA may influence cell–surface or cell–carrier interactions, highlighting the importance of application-specific optimization rather than direct one-to-one replacement of animal-derived BSA.
Applications: rBSA in IVD, diagnostics, and biotech workflows
Levprot’s recombinant albumin is designed to integrate seamlessly into established diagnostic and biotech workflows where BSA is routinely used as a blocking or stabilizing agent. By preserving core BSA protein functions, while delivering higher efficiency at lower working concentrations, Levprot rBSA is particularly well suited for in vitro diagnostics (IVD) and assay development environments.
In ELISA workflows, Levprot rBSA enables effective surface blocking while maintaining a stable balance between specific signal and background noise. This allows developers to reduce blocking agent concentration without compromising assay sensitivity, an advantage during both assay optimization and routine kit manufacturing. Similar benefits extend to CLIA assays, where consistent blocking performance and low non-specific binding are critical for signal reproducibility across batches.
For lateral flow and immunochromatographic tests, the use of animal-free rBSA supports better control over membrane saturation and reduces variability linked to raw material inconsistencies. This is particularly relevant in large-scale production, where lot-to-lot consistency directly impacts test performance and regulatory robustness.
Beyond diagnostics, Levprot’s recombinant albumin can be applied across a wide range of biotech workflows, including reagent formulation, routine protein handling, and cell culture systems where albumin plays a functional role as a carrier and stabilizing component. In cell culture applications, Levprot rBSA can be used as a defined, animal-free supplement or as part of FBS-reduction strategies, helping to replace undefined serum components while improving batch-to-batch consistency and regulatory alignment.
In these contexts, BSA alternatives are typically evaluated for performance, scalability, suitability for controlled manufacturing environments, and regulatory alignment. In all cases, adoption of Levprot rBSA requires minimal protocol changes, allowing teams to transition without disrupting existing, validated processes.
Implementation: how to apply rBSA while preserving BSA protein functions
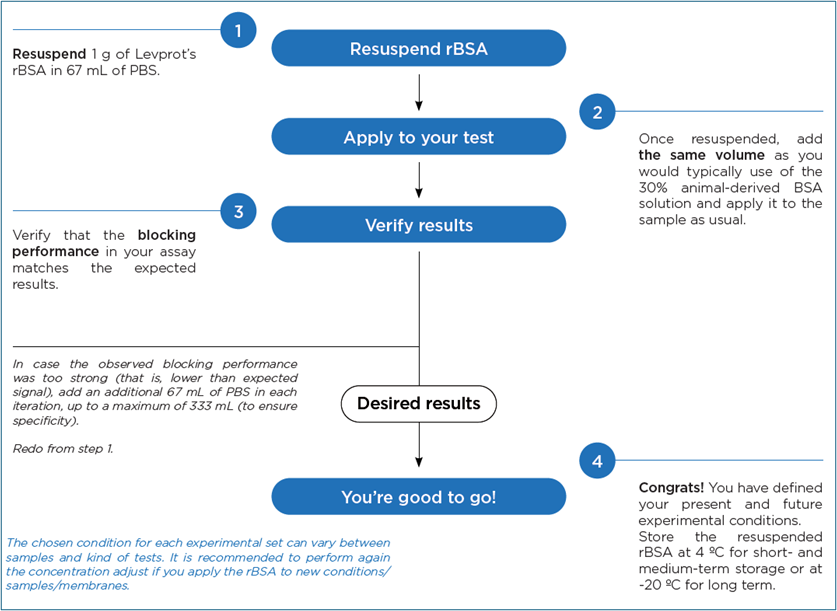
Implementation of Levprot rBSA is designed to be straightforward and compatible with existing laboratory and manufacturing workflows, while preserving core BSA protein functions. In diagnostic and immunoassay applications, the recombinant albumin is resuspended in standard buffers (e.g., PBS) and applied using the same volumes typically used for conventional animal-derived BSA solutions. Initial concentration adjustment is recommended during first-time use to define the optimal blocking conditions for each assay format.
Step-by-step workflow for implementing Levprot rBSA as a blocking or stabilizing agent in diagnostic and biotech applications.
Thanks to its higher functional efficiency, Levprot rBSA allows developers to progressively reduce the working concentration while maintaining equivalent specific signal and controlled non-specific background. This adjustment step is performed only once per application and does not require changes to assay design or downstream processing.
In cell culture and reagent formulation contexts, Levprot rBSA can be incorporated as a defined, animal-free component, supporting serum-reduction strategies while maintaining compatibility with established protocols. From a scale-up perspective, fermentation-based production and consistent purification ensure predictable supply and reproducibility across batches, facilitating technology transfer from R&D to routine manufacturing.
Levprot provides technical samples and application guidance to support first-time implementation.
Strategic view: precision fermentation proteins and animal-free BSA for regulated pipelines
The transition toward recombinant albumin solutions reflects a broader strategic shift within biotechnology and diagnostics: moving away from legacy, animal-derived raw materials toward designed, controlled, and regulatory-ready components. In this context, preserving BSA protein functions while improving consistency, scalability, and traceability is no longer a technical preference, but a structural requirement for modern development pipelines.
For biotech companies and diagnostics developers, preserving BSA protein functions while improving consistency and compliance represents a tangible competitive advantage. Levprot positions its recombinant albumin portfolio as part of a long-term platform built on precision fermentation proteins, addressing not only immediate performance needs but also future regulatory and manufacturing challenges.
By combining functional equivalence with animal-free origin and industrial scalability, Levprot enables organizations to standardize critical workflow components without compromising assay performance or development timelines. This approach supports robust and reproducible processes from early R&D through routine manufacturing, while remaining aligned with evolving regulatory expectations and market demands.
Comparison: Levprot rBSA vs conventional animal-derived BSA
| Parameter | Levprot rBSA | Conventional animal-derived BSA |
|---|---|---|
| Origin | Animal-free | Bovine plasma–derived |
| Production method | Precision fermentation | Serum fractionation |
| Batch consistency | High, controlled lot-to-lot reproducibility | Variable between batches |
| Functional yield | Higher effective performance per gram | Lower functional efficiency |
| Purity | High purity, homogeneous protein | High purity, residual serum components possible |
| Regulatory status | Animal-free, regulatory-ready | Requires animal-origin risk assessment |
| Scalability | Fermentation-based, predictable scale-up | Dependent on animal supply chains |
Conclusion: preserve BSA protein functions with animal-free rBSA
The shift toward recombinant albumin solutions reflects a clear evolution in biotech and diagnostics workflows. Preserving BSA protein functions while eliminating animal-derived variability enables more consistent, scalable, and regulatory-aligned processes across development and manufacturing. Levprot rBSA combines functional equivalence with animal-free origin and industrial readiness, supporting robust performance without disrupting established protocols.
Levprot provides technical samples, datasheets, and application support to help teams evaluate rBSA in their specific workflows. Contact us to discuss your application requirements or request samples.
FAQs (Frequently Asked Questions)
Can Levprot rBSA directly replace conventional BSA in immunoassays?
In most ELISA, CLIA, and related workflows, Levprot rBSA can be implemented using the same protocols, with an initial optimization step to define the optimal working concentration.
Does recombinant albumin preserve native BSA functionality?
Levprot rBSA is engineered to preserve essential BSA protein functions, including effective surface blocking, signal stabilization, and control of non-specific binding.
Is Levprot rBSA suitable for regulated environments?
Yes.
Its animal-free origin, batch consistency, and controlled production support use in regulated IVD and biotech applications.